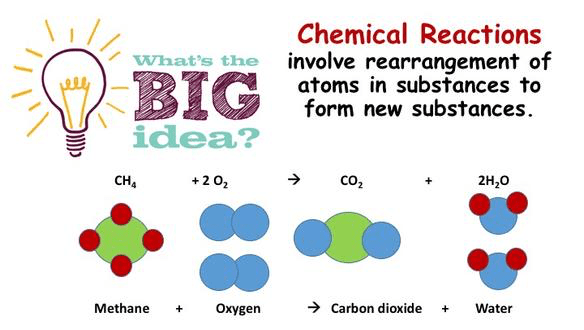Introduction
We know that particles react together when they bump into each other with enough energy (Collision theory). Up until know we have only looked at reactions that can only go in 1 direction. Once a match is struck it cannot reform, once a cake is baked you cannot go back to the original ingredients – These are irreversible reactions.
Now we look at reversible reactions – reactions that can go back to their starting materials.
The Essentials
Deeper Understanding
You can predict what will happen in a reaction if you alter the conditions. This is known as Le Chatelier’s Principle.
Concentration
If the concentration of a reactant is increased (A or B), more products will be formed until equilibrium is reached again. If the concentration of a product is decreased (C or D), more reactants will react until equilibrium is reached again.
Temperature
If the temperature of a reaction is increased (heated) the reaction move in the endothermic reaction (more C & D made). If the temperature of a reaction decreases (cooled) the reaction moves in the exothermic directions (more A & B made)
Pressure
If pressure is increased the reaction moves to where there are fewest gas molecules (A & B). If the pressure decreases it moves towards where there are more gas molecules (C & D)
Other Links
BBC Bitesize – Reversible Reactions
Review and Rate your Understanding
After revising the topic and completing the questions of the 100% sheet try the end of topic summary questions (answers given) and then print and complete the checklist to rate your understanding.
Chemistry_Separate_and_Trilogy_Rate_and_extent_chemical_change
Unit-6—The-rate-and-extent-of-chemical-change
Have you learnt all the facts on the 100% sheet?
Have you completed the BBC Bitesize tutorial?
Have you been able to complete all the questions on the 100% sheet?
Let us know how you feel about this topic in the comments section below. Any questions you have, just ask