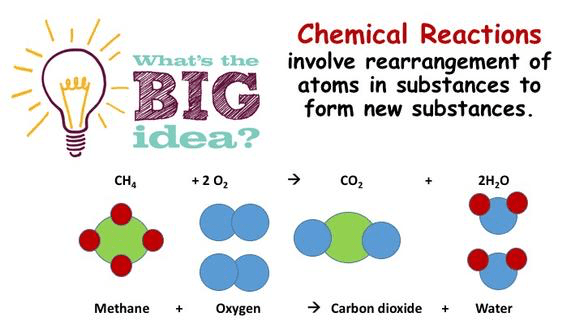Introduction
The Big Idea – Chemical reactions involve rearrangement of atoms in substances to form new substances.
We have looked at ionic compounds and can describe their properties and explain how they form. This topic is about using that knowledge to explore a range of chemical reactions. Using existing chemicals to make new molecules and compounds is what Chemistry and life is all about. Your body is carrying out billions of these reactions every second just to keep you alive.
Once again this topic builds on prior learning so if you you are not confident then visit the atomic structure page first and then the ionic compounds page before starting with this topic.
The Essentials
Acids
All acids contain hydrogen ions, all alkalis contain hydroxide ions. These ions combine to make water in a neutralisation reaction. The other ions in the acid and alkali combine to make the salt. You must be able to determine the name of the salt from the name of the acid and alkali
Making salts is a REQUIRED practical and you must learn the method and reasons for each step of the method. This is a very common exam question.
Summarise your knowledge using the worksheet
Metals
The method of metal extraction depends upon the reactivity of the metal. All metals, apart from the most unreactive (gold and silver) have combined with oxygen from the atmosphere to form metal oxides. This oxygen needs to be removed to form the cure metal. Carbon can be used for metals that are less reactive than carbon in a DISPLACEMENT reaction where carbon replaces the metal by combing with the oxide to form carbon dioxide and the pure metal. This movement of oxygen is an example of a Redox reaction. In the example above, carbon would be oxidised as it gains oxygen and the metal oxide would be reduced as it loses oxygen. If the metal is more reactive than carbon then electrolysis must be uses (see below).
Summarise your knowledge using the worksheet
Electrolysis
Electrolysis is using electricity to break ionic compounds apart into elements or molecules. It is just the reverse of how atoms become ions. The ions of the compound become atoms.
Positive ions will move towards the negative electrode where they will gin electrons to become neutral atoms and the negative ions will move towards the positive electrode and give up their extra electrons to the electrode to become neutral molecules. For ionic compounds the first part of the name (the metal) is ALWAYS the positive part.
Don’t PANIC about naming the electrodes, Positive in Anode and Negative Is Cathode.
Electrolysis only works with molten or dissolved ionic compounds as this is where the ions are free to move. You must know the difference between molten (pure melted liquid) and dissolved (water is present) ionic compounds and predict the movement of ions in each situation.
For dissolved compounds you must also consider the ions of water in electrolysis, H+ and OH- and follow these steps.
- List all 4 ions present
- Apply the following 2 rules on the 100% sheet to determine which of the ions are discharged (converted)
There is a REQUIRED practical involving electrolysis so learn the method and learn the gas tests so you could confirm the presence of any gas produced.
Deeper Understanding
Acids
For the higher paper you must be able to describe and explain the difference between strong and weak acids in terms of the degree of dissociation of the hydrogen ion into solution.
You must also know the reaction of acids with metals to produce a salt and hydrogen and describe these reactions in terms of loss or gain of electrons and Redox (OIL RIG)
Metals
For the higher paper you must be able to describe displacement reactions in terms of half equations and the loss or gain of electrons and Redox (OIL RIG)
Electrolysis
For the higher paper you must be able to write electron half equations including those for the hydrogen and hydroxide ions of water.
Other Links
Video links my nor work in school but will work at home.
BBC Bitesize – Chemical Changes
Exam Question Practice – gcsescienceteacher
Review and Rate your Understanding
After learning the content and practicing the questions on the 100% sheets, try the summary questions (answers included at the end) and them print and rate your understanding of the topic using the Unit 4 checklists below.
Chemistry_Trilogy_Chemical_changes
Unit-4—Chemical-Changes – Acids
Unit-4—Chemical Changes – Metals and REDOX
Unit-4—Chemical-Changes—Electrolysis
Have you learnt all the facts on the 100% sheet?
Have you completed the BBC Bitesize tutorial?
Have you been able to complete all the questions on the 100% sheet?
Let us know how you feel about this topic in the comments section below. Any questions you have, just ask.