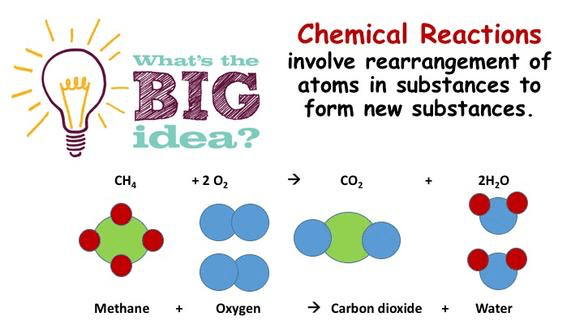Quantitative Chemistry-revisionIntroduction
The Big Idea – Chemical Reactions involve rearrangement of atoms in substances to form new substances.
In this topic we look at quantities. How much do we need to react to get a certain amount of product. If we want a defined amount of product, how much of each reactant do we need to start with. The maths of this is fairly simple but applying it to a chemistry context is more difficult. The more you practice these calculations the easier they will become.
The Essentials
You must know how to calculate formula masses from individual atomic masses (The top number from the periodic table). You get a mark for doing this in almost every calculation questions so make sure you always do this as a first step.
Remember that you are then dealing with ratios.
If 1 of A makes 3 of B then 2 of A makes 6 of B and 0.5 of A makes 1.5 B etc
The conservation of mass means that the entire sum of masses on the left hand side of an equations must equal to sum of masses on the right hand side.
In real life, this doesn’t always work out due to losing materials, incomplete reactions etc and so you can calculate a percentage yield (how much you got divided by how much you expected (maximum) X 100 )
Chemistry Cram 1.4 Quantitative Trilogy
Quantitative Chemistry-revision-flashcards
Summarise your knowledge using the worksheet
Deeper Understanding
The mole is simply a way of getting around the fact that different atoms have different masses, so if we simply used similar masses of everything we would actually have different numbers of each atom and that isn’t ideal for a chemist who wants to react known numbers of each atom together. To calculate a mole of something you simply take its mass in grammes and divide it by its molecular mass, 24g of carbon is 24/12 = 2 moles.
Once we know how many moles there are we can work out how many individual atoms there are by using the Avagadro number. In 1 mole of ANYTHING (atom, compound, molecule) there are 6.02 X10 to the power of 23. a very big number that shows how small individual atoms really are.
Other Links
BBC Bitesize – Quantitative Chemistry
Exam Question Practice – gcsescienceteacher
Review and Rate your Understanding
Try the questions below (answers given) then review your understanding
Chemistry_Trilogy_Quantitative_chemistry
Unit-3—Quantitative-Chemistry Checklist
Have you learnt all the facts on the 100% sheet?
Have you completed the BBC Bitesize tutorial?
Have you been able to complete all the questions on the 100% sheet?
Let us know how you feel about this topic in the comments section below. Any questions you have, just ask.