Introduction
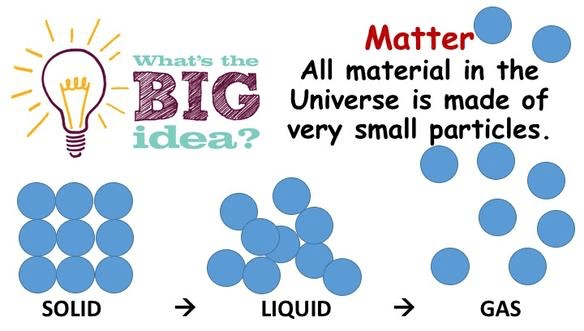
The Big Idea – All matter in the Universe is made up of very small particles.
Knowing the structure of the atom is not just an academic exercise. By knowing its structure we know how to use and manipulate it. We can explain the properties of all matter by understanding how proteins, neutrons and electrons move and interact. Without this knowledge the world around us would be a very different place. No microchips, no computers, nothing plastic, no mobile phones, no advanced medicines. Understand the atom and you’re well on your way to understanding the world around us.
This topic is divided into 3 parts.
- How did we arrive at our current understanding of the structure of the atom. Our idea of the atom has changed many times over the years and will change in the future
- How are all of these different atoms organised into a useful and usable format. How can we use this organisation known as the Periodic Table to understand the reactivity and properties of the different atoms known as elements.
- How does the structure of the atom relate to its properties and reactivity. Essential knowledge for predicting reactivity and linking to structure and properties of compounds which we explore later in the course.
The Essentials
This is THE ESSENTIAL TOPIC in Chemistry. EVERYTHING else you study in Chemistry is based upon this knowledge. This topic has been tested on EVERY exam paper. Do not leave this topic until you understand it. It will really help you with everything else in Chemistry.
The knowledge you absolutely need to know to make any sense of this topic
Chemistry Cram 1.1 Atomic Structure
Atomic Structure-revision-flashcards
Summarise your knowledge using the worksheet
Deeper Understanding
When you’re comfortable with the essentials think a little bit deeper and practice applying your knowledge
Use the timeline template or draw your own to review how and when our understanding of the structure of the atom changed. Who made the changes, what previous work did they build on.
The skills assessment is all about how the structure of the atom has changed. Here is a document to help you structure you report.
Help Sheet – Development of the Structure of the Atom
Why is it an important principle of Science that all discoveries are evidence based and that discoveries are published through a system of peer review?
Hydrogen is sometimes placed in group 1.
1) Describe a similarity and a difference between Hydrogen and the members of group 1.
2) Explain why Oxygen and Sulphur are in the same group even though Oxygen is a gas and sulphur is a solid.
3) Explain why Fluorine is the most reactive element in group 7
Other Links
BBC Bitesize – Atomic Structure
Video Tutorials – MyGCSE
Exam question practice – gcsescienceteacher
Review and Rate your Understanding
Try the questions below (answers given) then review your understanding
Chemistry_Separate_and_Trilogy_Atomic_structure_periodic_table
Unit-1—Atomic-structure-and-the-periodic-table-checklist
Have you learnt all the facts on the 100% sheet?
Have you completed the BBC Bitesize tutorial?
Have you been able to complete all the questions on the 100% sheet?
Let us know how you feel about this topic in the comments section below. Any questions you have, just ask.